Byrne, Robert, Diamond, Dermot  ORCID: 0000-0003-2944-4839, Fraser, Kevin J.
ORCID: 0000-0003-2944-4839, Fraser, Kevin J.  ORCID: 0000-0002-9718-5405, Izgorodina, Ekaterina, Forsyth, Maria and MacFarlane, Douglas R.
(2008)
Photo- and solvatochromic properties of nitrobenzospiropyran in ionic liquids containing the [NTf2]- anion.
Physical Chemistry Chemical Physics, 10
.
pp. 5919-5924.
ISSN 1463-9084
ORCID: 0000-0002-9718-5405, Izgorodina, Ekaterina, Forsyth, Maria and MacFarlane, Douglas R.
(2008)
Photo- and solvatochromic properties of nitrobenzospiropyran in ionic liquids containing the [NTf2]- anion.
Physical Chemistry Chemical Physics, 10
.
pp. 5919-5924.
ISSN 1463-9084
Abstract
The photo-, thermo- and solvatochromic properties of 2,3-dihydro-10,30,30-trimethyl-6-nitrospiro-
[1-benzopyran-2,20-1H-indole] (BSP-NO2) were studied in ILs containing the anion [NTf2]- by UV-Vis absorption spectroscopy, ab initio molecular orbital theory and density functional theory (DFT) calculations. It was found that the kinetics and thermodynamics of the BSP-NO2 MC (merocyanine) equilibrium was sensitive to the nature of the cation. It was also observed that the imidazolium cation can form a through-space orbital interaction with the MC isomer, rather than a simple electrostatic interaction, thus preventing the MC conversion back to the BSP-NO2 isomer. The BSP-NO2 MC equilibrium thus serves as a model system for studying modes of interaction of the cations in ionic liquids.
Metadata
| Item Type: | Article (Published) |
|---|---|
| Refereed: | Yes |
| Subjects: | Physical Sciences > Chemistry |
| DCU Faculties and Centres: | Research Institutes and Centres > CLARITY: The Centre for Sensor Web Technologies Research Institutes and Centres > National Centre for Sensor Research (NCSR) |
| Publisher: | Royal Society of Chemistry |
| Official URL: | http://dx.doi.org/10.1039/b806641h |
| Copyright Information: | © the Owner Societies 2008 |
| Use License: | This item is licensed under a Creative Commons Attribution-NonCommercial-Share Alike 3.0 License. View License |
| Funders: | Science Foundation Ireland |
| ID Code: | 14789 |
| Deposited On: | 07 Aug 2009 13:01 by Robert Byrne . Last Modified 18 Sep 2018 13:48 |
Documents
Full text available as:
Preview |
PDF
- Requires a PDF viewer such as GSview, Xpdf or Adobe Acrobat Reader
265kB |
![[thumbnail of b806641h-ga.gif]](https://doras.dcu.ie/14789/2.hassmallThumbnailVersion/b806641h-ga.gif) 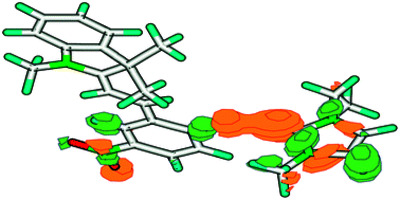 Preview |
Image (GIF)
19kB |
Downloads
Downloads
Downloads per month over past year
Archive Staff Only: edit this record